|
|
| Decretin |
Find this hormone for glucose regulation |
Decretins, hormones induced by fasting that suppress insulin production and secretion, have been postulated from classical human metabolic studies. From genetic screens, we identified Drosophila Limostatin (Lst), a peptide hormone that suppresses insulin secretion. Lst is induced by nutrient restriction in gut-associated endocrine cells. limostatin deficiency led to hyperinsulinemia, hypoglycemia, and excess adiposity. A conserved 15-residue polypeptide encoded by limostatin suppressed secretion by insulin-producing cells. Targeted knockdown of CG9918, a Drosophila ortholog of Neuromedin U receptors (NMURs), in insulin-producing cells phenocopied limostatin deficiency and attenuated insulin suppression by purified Lst, suggesting CG9918 encodes an Lst receptor. NMUR1 is expressed in islet β cells, and purified NMU suppresses insulin secretion from human islets. A human mutant NMU variant that co-segregates with familial early-onset obesity and hyperinsulinemia fails to suppress insulin secretion. We propose Lst as an index member of an ancient hormone class called decretins, which suppress insulin output.
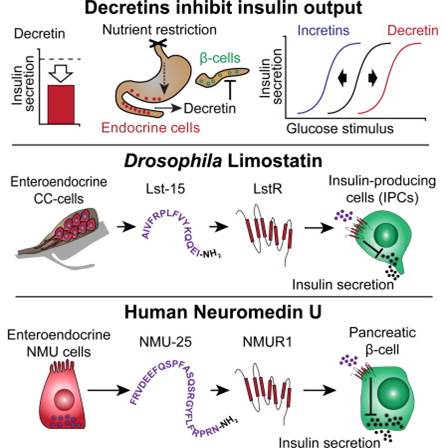
Alfa RW, Park S, Skelly KR et al., Cell Metab. 2015 Feb 3;21(2):323-33. doi: 10.1016/j.cmet.2015.01.006.
As a master controller of glucose metabolism, insulin is a tightly regulated hormone. Several hormones, referred to as incretins, enhance insulin secretion in response to increased glucose; however, few endocrine hormones have been identified that reduce insulin secretion in response to prolonged starvation. Alfa et al. ectopically expressed selected starvation-regulated genes in Drosophila and identified CG8317, which the authors named limostatin (lst) for the Greek goddess of starvation, as one that caused phenotypes associated with insulin deficiency. The abundance of lst mRNA peaked in adult flies after 24 hours of starvation and was reduced by refeeding the flies carbohydrates, but not protein. The lst gene encodes a putative propeptide with a signal peptide (for secretion) and several dibasic cleavage sites. Two arginine residues in the dibasic cleavage sites were necessary for the insulin-deficiency phenotypes associated with overexpression. Flies with a P element insertion that eliminated lst (lst1) had phenotypes associated with excess insulin, such as hypoglycemia, obesity, and shortened lifespan, and these flies also had increased abundance of a tagged form of circulating insulin-like peptide 2 (Ilp2). Silencing the electrical activity of insulin-producing cells (IPCs, the fly equivalent of pancreatic β cells), which is necessary for Ilp secretion, eliminated differences in triglyceride content between control and lst1 flies. Analysis of flies expressing an lstpromoter reporter indicated that Lst was produced by gut-associated endocrine cells that also produce an incretin hormone. Flies in which lst was knocked down in these cells had a similar phenotype as lst1 flies, and expression oflst specifically in those cells rescued the lst1 phenotype. Application of a 15-amino-acid peptide derived from Lst, Lst-15, inhibited calcium signals in and inhibited Ilp2 release from IPCs. Screening for genes encoding G protein–coupled receptors (GPCRs), which are commonly targets of peptide hormones, that exhibited increased expression inlst1 and decreased expression when lst was overexpressed identified CG9918. IPC-specific knockdown of CG9918phenocopied lst1 and blocked the attenuating effect of Lst-15 on Ilp2 secretion from IPCs. The GPCRs most similar to CG9918 in mammals are the neuromedin U receptors (NMURs), and NMUR1 is found in the periphery. NMUR1 protein and mRNA were present in human pancreatic β cells and the gene encoding the ligand NMU was expressed in stomach and duodenum (human foregut, analogous to the cells in flies that produced Lst). Application of NMU-25 to human islets inhibited glucose-stimulated insulin release and a mutant form of NMU associated with obesity failed to suppress insulin release. Thus, Lst-15 and NMU-25 appear to function as endocrine decretin hormones.
Auther: Nancy R. Gough, Sci. Signal., 10 February 2015 Vol. 8, Issue 363, p. ec29
Neuronostatin is a recently described peptide hormone encoded by the somatostatin gene. We previously showed that intraperitoneal injection ofneuronostatin into mice resulted in c-Jun accumulation in pancreatic islets in a pattern consistent with the activation of glucagon-producing α-cells. We therefore hypothesized that neuronostatin could influence glucose homeostasis via a direct effect on the α-cell. Neuronostatin enhanced low-glucose-induced glucagon release in isolated rat islets and in the immortalized α-cell line αTC1-9. Furthermore, incubation with neuronostatin led to an increase in transcription of glucagon mRNA, as determined by RT-PCR. Neuronostatin also inhibited glucose-stimulated insulin secretion from isolated islets. However, neuronostatin did not alter insulin release from the β-cell line INS 832/13, indicating that the effect of neuronostatin on insulin secretion may be secondary to a direct action on the α-cell. In agreement with our in vitro data, intra-arterial infusion of neuronostatin in male rats delayed glucose disposal and inhibited insulin release during a glucose challenge. These studies suggest that neuronostatin participates in maintaining glucose homeostasis through cell-cell interactions between α-cells and β-cells in the endocrine pancreas, leading to attenuation in insulin secretion.
Salvatori AS, Elrick MM, Samson WK et al., Am J Physiol Endocrinol Metab. 2014 Jun 1;306(11):E1257-63. doi: 10.1152/ajpendo.00599.2013. Epub 2014 Apr 15.
Neuromedin U (NMU) are bioactive peptides with a common C-terminal heptapeptide sequence (FLFRPRN-amide, 1a) among mammals, which is responsible for receptor activation, namely NMU receptor types 1 (NMUR1) and 2 (NMUR2). Among the various physiological actions of NMU, the anorexigenic effect has recently attracted attention in drug discovery efforts for treating obesity. Although several structure-activity relationship (SAR) studies have been reported, receptor-selective small peptide agonists have yet to be disclosed. Herein a SAR study of 1a-derived peptide derivatives is described. We initially screened both human NMUR1- and NMUR2-selective peptides in calcium-mobilization assays with cells transiently expressing receptors. Then we performed a precise assay with a stable expression system of receptors and consequently discovered hexapeptides 8d and 6b possessing selective agonist activity toward each respective receptor. Hexapeptide 6b, which selectively activates NMUR2 without significant NMUR1 activation, should aid in the development of anorexigenic drugs as well as advance NMU-related endocrinological research.
Takayama K, Mori K, Taketa K et al., J Med Chem. 2014 Aug 14;57(15):6583-93. doi: 10.1021/jm500599s. Epub 2014 Jul 18.
The interaction of glucagon-like peptide-I (GLP-I) and galanin in clonal endocrine pancreatic cells was characterized. By Northern blot analysis the presence of GLP-I receptor mRNA was shown in B (beta TC-1 cells) and D (RIN 1048-38) cells but not in A (INR1 G9) cells, thus confirming functional data demonstrating the absence of active GLP-I receptors on glucagon-producing cells. Galanin receptors were detected on B and D cells but not on A cells. In B and D cells galanin inhibited the GLP-I stimulated adenylate cyclase activity. Treatment of insulin- and somatostatin-producing cells with GLP-I increased intracellular cAMP levels, and this was dampened by galanin, GLP-I stimulated the activity of protein kinase A in B and D cells, which was also inhibited by galanin. Galanin alone did not influence B- and D-cell function. These data show that in the endocrine pancreas B and D cells but not A cells express GLP-I and galanin receptors. The interaction of GLP-I and galanin might act in the endocrine pancreas as a physiological inhibitor of the potent incretin hormone GLP-I. Therefore, we suggest galanin is a 'decretin'.
Fehmann HC, Janssen M, Göke B., Acta Diabetol. 1995 Oct;32(3):176-81.
|
|
|
%046-33%%;046-30%;%026-08%;%neuromedin u%
|
|
|


