|
|
|
Bioactive Human Relaxin-2 in Bulk Quantity Available |
Examples of Phoenix’s Relaxin-2 Bioactivity in Literatures
| Relaxin improves TNF-α-induced endothelial dysfunction: the role of glucocorticoid receptor and phosphatidylinositol 3-kinase signalling |
Dschietzig T |
Cardiovasc Res. 2012 ;95(1):97-107. doi: 10.1093/cvr/cvs149 |
Immundiagnostik AG, Stubenwaldallee 8a, Bensheim, Germany. |
improves endothelial function by inhibiting vascular inflammation |
| Relaxin 2 is functional at the ocular surface and promotes corneal wound healing |
Hampel U |
Invest Ophthalmo Vis Sci. 2012 ;53(12):7780-7790. doi: 10.1167/iovs.12-10714 |
Department of Anatomy II, Friedrich Alexander University of Erlangen-Nürnberg, Erlangen, Germany. |
Improves cell proliferation, migration and re-epithelialzation of corncal wounds |
| Relaxin reduces xenograft tumour growth of human MDA-MB-231 breast cancer cells |
Y Radestock |
Breast Cancer Research 2008, 10:1-15. doi:10.1186/bcr2136 |
Clinics of General, Visceral and Vascular Surgery, Magdeburger Str. 18, Martin-Luther-University Halle Wittenberg, 06097 Halle, Germany. |
increases cell motility dose-dependently |
| In Vitro Pharmacological Characterization of RXFP3 Allosterism: An Example of Probe Dependency |
Alvarez-Jaimes L |
PLoS One. 2012;7(2):e30792. doi: 10.1371/journal.pone.0030792. Epub 2012 Feb 7. |
Janssen Pharmaceutical Companies of Johnson & Johnson, San Diego, California, United States of America |
cAMP accumulation |
| Effect of Relaxin on Human Sperm Functions and Fertilizing Ability |
Ferlin A |
Journal of Andrology, (33), 3, 474-80, 2012 |
University of padova, Padova, Italy |
increase in sperm hyperactivation, intracellular calcium and cAMP, and induce acrosome reaction |
| Effect of relaxin on myocardial ischemia injury induced by isoproterenol |
Zhang J |
Peptides. 2005 Sep;26(9):1632-1639 |
Institute of Cardiovascular Research, Peking University , China |
Pretect from myocardial ischemia injury and fibrosis |
| Intracellular signaling pathways involved in the relaxin-induced proliferation of rat Sertoli cells. |
Nascimento A R |
Eur J Pharmacol. 2012 Sep 15;691(1-3):283-91. doi: 10.1016/j.ejphar.2012.07.021 |
Universidade Federal de São Paulo, São Paulo, Brazil. |
Induced Sertoli cell proliferation, and activation the activity of ERK1/2 phosphorylation and G(i) |
| Relaxin prevents the development of severe acute pancreatitis |
Laura Iris Cosen-Binker |
World J Gastroenterol 2006 March 14;12(10):1558-1568; |
RHC-LICB Medical Research Institute-Buenos Aires, Argentina |
prevents the development of severe acute pancreatitis |
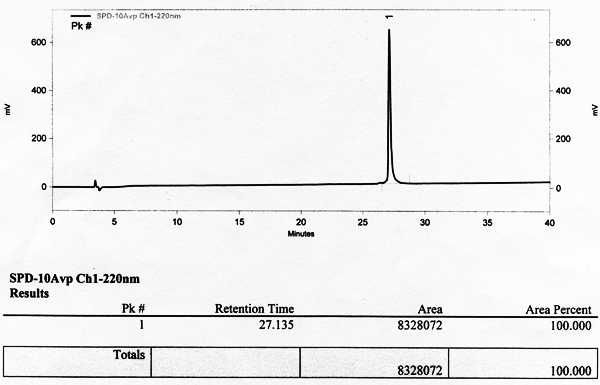
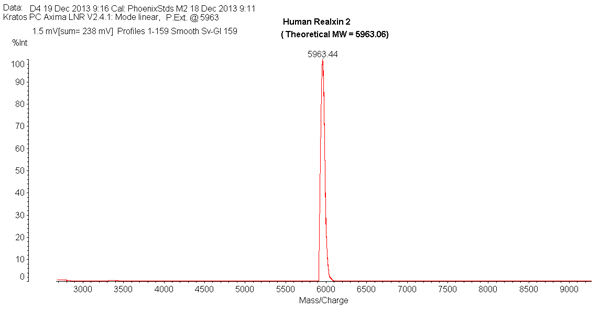
Human relaxin-2 (hereafter simply defined as "relaxin") is a 6-kDa peptidic hormone best known for the physiological role played during pregnancy in the growth and differentiation of the reproductive tract and in the renal and systemic hemodynamic changes. This factor can also be involved in the pathophysiology of arterial hypertension and heart failure, in the molecular pathways of fibrosis and cancer, and in angiogenesis and bone remodeling. It belongs to the relaxin peptide family, whose members comprehensively exert numerous effects through interaction with different types of receptors, classified as relaxin family peptide (RXFP) receptors (RXFP1, RXFP2, RXFP3, RXFP4). Research looks toward the in-depth examination and complete understanding of relaxin in its various pleiotropic actions. The intent is to evaluate the likelihood of employing this substance for therapeutic purposes, for instance in diseases where a deficit could be part of the underlying pathophysiological mechanisms, also avoiding any adverse effect. Relaxin is already being considered as a promising drug, especially in acute heart failure. A careful study of the different RXFPs and their receptors and the comprehension of all biological activities of these hormones will probably provide new drugs with a potential wide range of therapeutic applications in the near future.
Cernaro V, Lacquaniti A, Lupica R, et al., Med Res Rev. 2014 Jan;34(1):77-105. doi: 10.1002/med.21277. Epub 2013 Feb 11.
AIMS: Serelaxin (Relaxin 2) is effective in relieving dyspnoea and improving multiple outcomes in acute heart failure (AHF). Many AHF patients have preserved ejection fraction (HFpEF). Given the lack of evidence-based therapies in this population, we evaluated the effects of serelaxin according to EF in RELAX-AHF trial.
METHODS AND RESULTS: RELAX-AHF randomized 1161 AHF patients to 48-h serelaxin (30 μg/kg/day) or placebo within 16 h from presentation. We compared the effects of serelaxin on efficacy endpoints, safety endpoints, and biomarkers of organ damage between preserved (≥50%) and reduced (<50%, HFrEF) EF. HFpEF was present in 26% of patients. Serelaxin induced a similar dyspnoea relief in HFpEF vs. HFrEF patients by visual analogue scale-area under the curve (VAS-AUC) through Day 5 [mean change, 461 (-195, 1117) vs. 397 (10, 783) mm h, P = 0.87], but had possibly different effects on the proportion of patients with moderately or markedly dyspnoea improvement by Likert scale at 6, 12, and 24 h [odds ratio for favourable response, 1.70 (0.98, 2.95) vs. 0.85 (0.62, 1.15), interaction P = 0.030]. No differences were encountered in the effect of serelaxin on short- or long-term outcome between HFpEF and HFrEF patients including cardiovascular death or hospitalization for heart/renal failure through Day 60, cardiovascular death through Day 180, and all-cause death through Day 180. Similar safety and changes in biomarkers (high-sensitivity troponin T, cystatin-C, and alanine/aspartate aminotransferases) were found in both groups.
CONCLUSIONS: In AHF patients with HFpEF compared with those with HFrEF, serelaxin was well tolerated and effective in relieving dyspnoea and had a similar effect on short- and long-term outcome, including survival improvement.
Filippatos G, Teerlink JR, Farmakis D et al., Eur Heart J. 2013 Dec 6. [Epub ahead of print]
INTRODUCTION: Early allograft dysfunction following lung transplantation is mainly an ischemia/reperfusion (IR) injury. We showed that relaxin-2 (relaxin, Phoenix Pharmaceuticals) exerts a protective effect in lung IR, attributable to decreases in endothelin-1 (ET-1) production, leukocyte recruitment, and free radical generation. Here, we summarize our investigations into relaxin's signalling.
MATERIALS AND METHODS: Isolated rat lungs were perfused with vehicle or 5 nM relaxin (n=6-10 each). Thereafter, experiments were conducted in the presence of relaxin plus vehicle, the protein kinase A inhibitors H-89 and KT-5720, the NO synthase (NOS) inhibitor L-NAME, the iNOS inhibitor 1400W, the nNOS inhibitor SMTC, the extracellular signal-regulated kinase-1/2 (ERK-1/2) inhibitor PD-98059, the phosphatidylinositol-3 kinase (PI3K) inhibitor wortmannin,the endothelin type-B (ETB) antagonist A-192621, or the glucocorticoid receptor (GR) antagonist RU-486. After 90 min ischemia and 90 min reperfusion we determined wet-to-dry (W/D) weight ratio, mean pulmonary arterial pressure (MPAP), vascular release of ET-1, neutrophil elastase (NE), myeloperoxidase (MPO), and malondialdehyde (MDA). Primary rat pulmonary vascular cells were similarly treated.
RESULTS: IR lungs displayed significantly elevated W/D ratios, MPAP, as well as ET-1, NE, MDA, and MPO. In the presence of relaxin, all of these parameters were markedly improved. This protective effect was completely abolished by L-NAME, 1400W, PD-98059, and wortmannin whereas neither PKA and nNOS inhibition nor ETB and GR antagonism were effective. Analysis of NOS gene expression and activity revealed that the relaxin-induced early and moderate iNOS stimulation is ERK-1/2-dependent and counter-balanced by PI3K. Relaxin-PI3K-related phosphorylation of a forkhead transcription factor, FKHRL1, paralleled this regulation. In pulmonary endothelial and smooth muscle cells, FKHRL1 was essential to relaxin-PI3K signalling towards iNOS.
CONCLUSION: In this short-time experimental setting, relaxin protects against IR-induced lung injury via early and moderate iNOS induction, dependent on balanced ERK-1/2 and PI3K-FKHRL1 stimulation. These findings render relaxin a candidate drug for lung preservation.
Alexiou K, Wilbring M, Matschke K, Dschietzig T., PLoS One. 2013 Sep 30;8(9):e75592. doi: 10.1371/journal.pone.0075592.
AIMS: Human relaxin-2 influences renal and cardiovascular functions. We investigated its effects on experimental endothelial dysfunction.
METHODS AND RESULTS: Acetylcholine-mediated vasodilation of rat aortic rings, impaired by 48 h tumour necrosis factor-α (TNF-α) treatment, was dose-dependently improved by relaxin ( Phoenix Pharmaceuticals) co-incubation, an effect sensitive to phosphatidylinositol 3-kinase (PI3K) inhibition and the glucocorticoid receptor (GR) antagonist RU-486. TNF increased endothelial nitric oxide synthase (eNOS) phosphorylation at Thr495 and decreased total eNOS expression and both basal and stimulated eNOS activity. Relaxin co-incubation did not affect eNOS expression but improved its activity via PI3K-dependent Thr495 dephosphorylation and Ser1177 phosphorylation, and additional Ser633 phosphorylation. Via GR, relaxin attenuated the TNF-related stimulation of endothelin-1 expression, superoxide and nitrotyrosine formation, and arginase II expression. Relaxin restored, via GR-CCAAT/enhancer-binding protein-β (c/EBP-β)-mediated promoter stimulation, the compromised expression of superoxide dismutase-1 (SOD1). In rat aortic endothelial cells, relaxin activated protein kinase B (Akt) and repressed TNF-induced nuclear factor-κB and activator protein-1. Finally, the relevance of the different findings to the model used was proved by pharmacological interventions.
CONCLUSION: Relaxin improved endothelial dysfunction by promoting eNOS activity, suppressing endothelin-1 and arginase-II expression, and up-regulating SOD1 via GR, GR-c/EBP-β, and PI3K-Akt pathways. This corroborates the notion that it functions as an endogenous and potentially therapeutic vasoprotector.
Dschietzig T, Brecht A, Bartsch C et al., Cardiovasc Res. 2012 Jul 1;95(1):97-107. doi: 10.1093/cvr/cvs149. Epub 2012 Apr 16.
Regulation of Sertoli cell number is a key event to determine normal spermatogenesis. We have previously shown that relaxin and its G-protein coupled receptor RXFP1 are expressed in rat Sertoli cells, and that relaxin stimulates Sertoli cell proliferation. This study examined the mechanisms underlying the mitogenic effect of relaxin in a primary culture of Sertoli cells removed from testes of immature rats. Stimulation with exogenous relaxin increased Sertoli cell number and the expression of the proliferating cell nuclear antigen (PCNA), but did not affect the mRNA level of the differentiation markers cadherins 1 and 2. Relaxin-induced Sertoli cell proliferation was blocked by inhibition of MEK/ERK1/2 or PI3K/AKT pathways, but not by inhibition of PKC or EGFR activity. Relaxin induced a rapid and transient activation of ERK1/2 phosphorylation, which was MEK and SRC-dependent, and involved upstream activation of G(i). AKT activation could be detected 5 min after relaxin stimulation, and was still detected after 24h of stimulation with relaxin. Relaxin-induced AKT phosphorylation was G(i)- but not PKA-dependent, and it was blocked by both PI3K and MEK inhibitors. In conclusion, the mitogenic effect of relaxin in Sertoli cell involves coupling to G(i) and activation of both MEK/ERK1/2 and PI3K/AKT pathways.
Nascimento AR, Pimenta MT, Lucas TF, et al., Eur J Pharmacol. 2012 Sep 15;691(1-3):283-91. doi: 10.1016/j.ejphar.2012.07.021. Epub 2012 Jul 20.
Relaxin is a circulating hormone with functions in pregnancy, parturition, and other aspects of female reproduction. It is also secreted from the prostate gland into the seminal fluid; however, the role of relaxin in male reproduction is debated. Studies conducted in the past have suggested possible actions on human spermatozoa, but the data were contrasting. Here, we show that the relaxin receptor RXFP1 (Relaxin Family Peptide Receptor 1) is expressed in human spermatozoa, and it mainly localizes in the astrodome. In vitro studies on human sperm demonstrated that this hormone attenuates the natural decline in sperm motility and maintains higher mitochondrial activity and lower apoptosis level. Furthermore, relaxin induced an increase in sperm hyperactivation, intracellular calcium and cAMP, and acrosome reaction. These effects were abolished by the use of the specific anti-RXFP1 antibody. Relaxin concentrations were low in the blood (x ± SD, 0.16 ± 0.03 nM) and very high in the seminal plasma (x ± SD, 10.3 ± 4.0 nM), confirming its secretion mainly by the prostate. Taken together, these data demonstrate that relaxin influences positively many sperm functions linked to fertilizing ability, and it preserves sperm functionality, with possible practical value in assisted reproduction techniques.
Ferlin A, Menegazzo M, Gianesello L, et al., J Androl. 2012 May-Jun;33(3):474-82. doi: 10.2164/jandrol.110.012625. Epub 2011 Sep 8.
The omnipresent 6-kDa polypeptide relaxin (RLX) is emerging as a multifunctional endocrine and paracrine factor in a broad range of target tissues including cardiovascular tissues. To explore the pathophysiological roles of RLX in ischemic cardiovascular diseases, we studied the changes in RLX mRNA level in the myocardium and the effect of RLX supplements in rats with isoproterenol (ISO)-induced myocardial injury. In ISO-treated rats, RLX levels in myocardia and plasma increased 3.7- and 6.9-fold, respectively (P<0.01), the mRNA level increased significantly in myocardia compared with controls. Co-administration of RLX (0.2 and 2.0 microg/kg/d) and ISO increased left-ventricular pressure development and decreased left ventricular end-diastolic pressure (LVDEP) (all P<0.01). Malondialdehyde content in myocardia and lactate dehydrogenase and creatine phosphokinase activities in plasma in RLX-treated rats decreased markedly compared with that in ISO-treated alone rats (P<0.01 or P<0.05). In the high-dose RLX group, fibroblastic hyperplasia was relieved in myocardia, hydroxyproline level was lower, by 33% (P<0.05), and endothelin content in plasma was lower, by 31% (P<0.01) than in the ISO-alone group. Compared with control group, any indexes in sham rats treated with high-dose RLX were unaltered (all P>0.05). These results showed an up-regulation of myocardial RLX during ISO-induced myocardial ischemia injury and the protective effect of RLX on ISO-induced cardiac inhibition and fibrosis, which suggests that RLX could be an endogenous cardioprotective factor in ischemic heart diseases.
Zhang J, Qi YF, Geng B, et al., Peptides. 2005 Sep;26(9):1632-9.
AIM:
To investigate the severity of acute pancreatitis (AP) is associated to the intensity of leukocyte activation, inflammatory up-regulation and microcirculatory disruption associated to ischemia-reperfusion injury. Microvascular integrity and inhibition of pro-inflammatory mediators are key-factors in the evolution of AP. Relaxin is an insulin-like hormone that has been attributed vasorelaxant properties via the nitric oxide pathway while behaving as a glucocorticoid receptor agonist.
METHODS:
AP was induced by the bilio-pancreatic duct-outlet-exclusion closed-duodenal-loops model. Treatment with relaxin was done at different time-points. Nitric oxide synthase inhibition by L-NAME and glucocorticoid receptor (GR) blockage by mifepristone was considered. AP severity was assessed by biochemical and histopathological analyses.
RESULTS:
Treatment with relaxin reduced serum amylase, lipase, C-reactive protein, IL-6, IL-10, hsp72, LDH and 8-isoprostane as well as pancreatic and lung myeloperoxidase. Acinar and fat necrosis, hemorrhage and neutrophil infiltrate were also decreased. ATP depletion and ADP/ATP ratio were reduced while caspases 2-3-8 and 9 activities were increased. L-NAME and mifepristone decreased the efficiency of relaxin.
CONCLUSION:
Relaxin resulted beneficial in the treatment of AP combining the properties of a GR agonist while preserving the microcirculation and favoring apoptosis over necrosis.
|
|
|
relaxin 2
%relaxin 2%
|
|
|


