The role of skeletal muscle in nonshivering thermogenesis (NST) is not well understood. Here we show that sarcolipin (Sln), a newly identified regulator of the sarco/endoplasmic reticulum Ca(2+)-ATPase (Serca) pump, is necessary for muscle-based thermogenesis. When challenged to acute cold (4 °C), Sln(-/-) mice were not able to maintain their core body temperature (37 °C) and developed hypothermia. Surgical ablation of brown adipose tissue and functional knockdown of Ucp1 allowed us to highlight the role of muscle in NST. Overexpression of Sln in the Sln-null background fully restored muscle-based thermogenesis, suggesting that Sln is the basis for Serca-mediated heat production. We show that ryanodine receptor 1 (Ryr1)-mediated Ca(2+) leak is an important mechanism for Serca-activated heat generation. Here we present data to suggest that Sln can continue to interact with Serca in the presence of Ca(2+), which can promote uncoupling of the Serca pump and cause futile cycling. We further show that loss of Sln predisposes mice to diet-induced obesity, which suggests that Sln-mediated NST is recruited during metabolic overload. These data collectively suggest that SLN is an important mediator of muscle thermogenesis and whole-body energy metabolism.
Bal N.C. et al., Nat Med. 2012 Sep 9;18(10):1575-9. doi: 10.1038/nm.2897. Epub 2012 Sep 9.
Sarcolipin (SLN) is a key regulator of sarco(endo)plasmic reticulum (SR) Ca(2+)-ATPase (SERCA), and its expression is altered in diseased atrial myocardium. To determine the precise role of SLN in atrial Ca(2+) homeostasis, we developed a SLN knockout (sln-/-) mouse model and demonstrated that ablation of SLN enhances atrial SERCA pump activity. The present study is designed to determine the long-term effects of enhanced SERCA activity on atrial remodeling in the sln-/- mice. Calcium transient measurements show an increase in atrial SR Ca(2+) load and twitch Ca(2+) transients. Patch-clamping experiments demonstrate activation of the forward mode of sodium/calcium exchanger, increased L-type Ca(2+) channel activity, and prolongation of action potential duration at 90% repolarization in the atrial myocytes of sln-/- mice. Spontaneous Ca(2+) waves, delayed afterdepolarization, and triggered activities are frequent in the atrial myocytes of sln-/- mice. Furthermore, loss of SLN in atria is associated with increased interstitial fibrosis and altered expression of genes encoding collagen and other extracellular matrix proteins. Our results also show that the sln-/- mice are susceptible to atrial arrhythmias upon aging. Together, these findings indicate that ablation of SLN results in increased SERCA activity and SR Ca(2+) load, which, in turn, could cause abnormal intracellular Ca(2+) handling and atrial remodeling.
Xie LH, Shanmugam M, Park JY, Zhao Z, Wen H, Tian B, Periasamy M, Babu GJ. Am J Physiol Cell Physiol. 2012 Jun;302(12):C1762-71. Epub 2012 Apr 11.
The pathophysiological mechanisms of both familial and sporadic Amyotrophic Lateral Sclerosis (ALS) are unknown, although growing evidence suggests that skeletal muscle tissue is a primary target of ALS toxicity. Skeletal muscle biopsies were performed on transgenic SOD1G93A mice, a mouse model of ALS, to determine genetic biomarkers of disease longevity. Mice were anesthetized with isoflurane, and three biopsy samples were obtained per animal at the three main stages of the disease. Transcriptional expression levels of seventeen genes, Ankrd1, Calm1, Col19a1, Fbxo32, Gsr, Impa1, Mef2c, Mt2, Myf5, Myod1, Myog, Nnt, Nogo A, Pax7, Rrad, Sln and Snx10, were tested in each muscle biopsy sample. Total RNA was extracted using TRIzol Reagent according to the manufacturer's protocol, and variations in gene expression were assayed by real-time PCR for all of the samples. The Pearson correlation coefficient was used to determine the linear correlation between transcriptional expression levels throughout disease progression and longevity. Consistent with the results obtained from total skeletal muscle of transgenic SOD1G93A mice and 74-day-old denervated mice, five genes (Mef2c, Gsr, Col19a1, Calm1 and Snx10) could be considered potential genetic biomarkers of longevity in transgenic SOD1G93A mice. These results are important because they may lead to the exploration of previously unexamined tissues in the search for new disease biomarkers and even to the application of these findings in human studies.
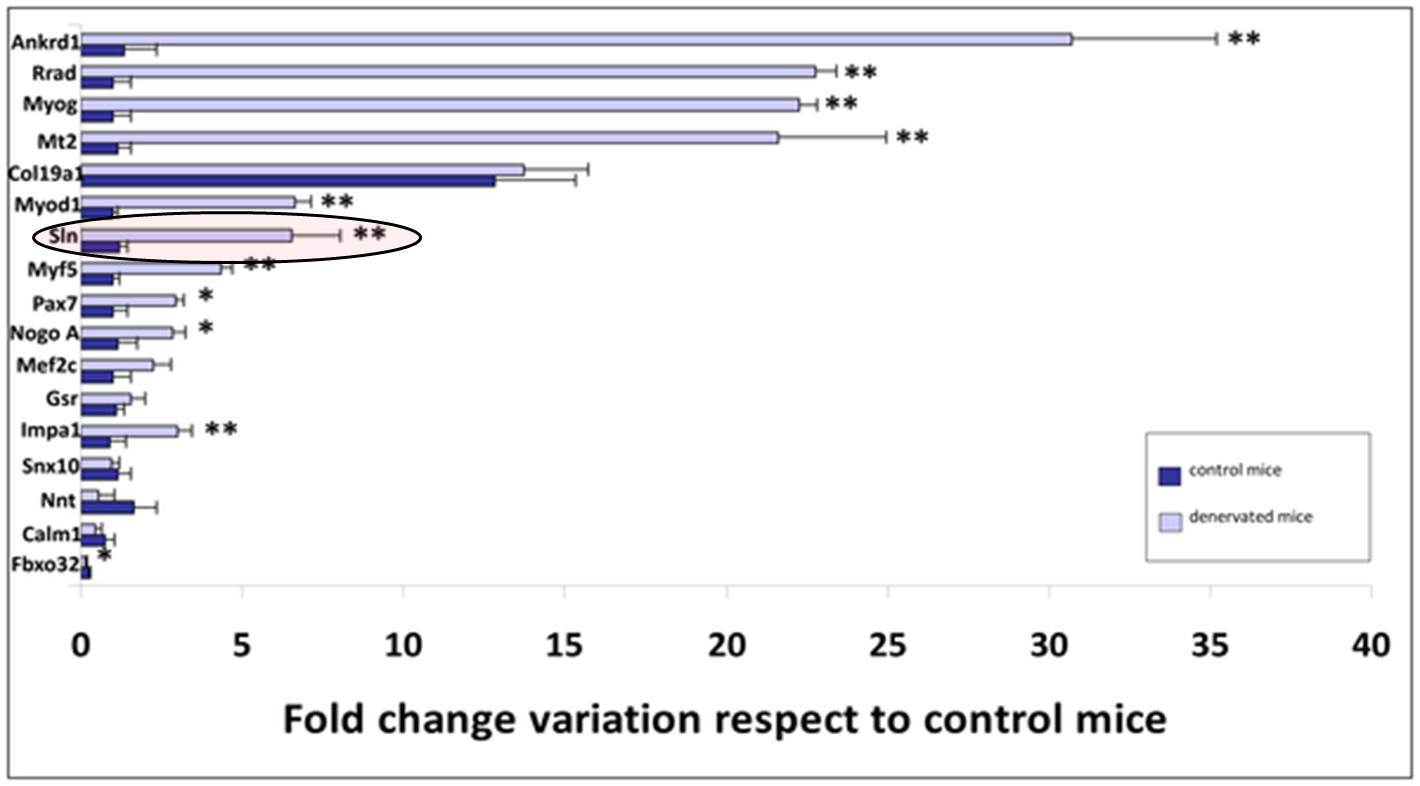
Calvo AC, Manzano R, Atencia-Cibreiro G, Oliván S, et al. (2012) PLoS ONE 7(3): e32632. doi:10.1371/journal.pone.0032632
Sarcolipin (SLN), a key regulator of cardiac sarco(endo)plasmic reticulum (SR) Ca(2+) ATPase, is predominantly expressed in atria and mediates β-adrenergic responses. Studies have shown that SLN mRNA expression is decreased in human chronic atrial fibrillation (AF) and in aortic banded mouse atria; however, SLN protein expression in human atrial pathology and its role in atrial SR Ca(2+) uptake are not yet elucidated. In the present study, we determined the expression of major SR Ca(2+) handling proteins in atria of human AF patients and in human and in a mouse model of heart failure (HF). We found that the expression of SR Ca(2+) uptake and Ca(2+) release channel proteins are significantly decreased in atria but not in the ventricles of pressure-overload induced HF in mice. In human AF and HF, the expression of SLN protein was significantly decreased; whereas the expressions of other major SR Ca(2+) handling proteins were not altered. Further, we found that the SR Ca(2+) uptake was significantly increased in human AF. The selective downregulation of SLN and enhanced SR Ca(2+) uptake in human AF suggest that SLN downregulation could play an important role in abnormal intracellular Ca(2+) cycling in atrial pathology.
Shanmugam M. et. al, Biochem Biophys Res Commun. 2011 Jun 24;410(1):97-101. Epub 2011 May 25.
Sarcolipin is a novel regulator of cardiac sarcoplasmic reticulum Ca2+ ATPase 2a (SERCA2a) and is expressed abundantly in atria. In this study we investigated the physiological significance of sarcolipin in the heart by generating a mouse model deficient for sarcolipin. The sarcolipin-null mice do not show any developmental abnormalities or any cardiac pathology. The absence of sarcolipin does not modify the expression level of other Ca2+ handling proteins, in particular phospholamban, and its phosphorylation status. Calcium uptake studies revealed that, in the atria, ablation of sarcolipin resulted in an increase in the affinity of the SERCA pump for Ca2+ and the maximum velocity of Ca2+ uptake rates. An important finding is that ablation of sarcolipin resulted in an increase in atrial Ca2+ transient amplitudes, and this resulted in enhanced atrial contractility. Furthermore, atria from sarcolipin-null mice showed a blunted response to isoproterenol stimulation, implicating sarcolipin as a mediator of β-adrenergic responses in atria. Our study documented that sarcolipin is a key regulator of SERCA2a in atria. Importantly, our data demonstrate the existence of distinct modulators for the SERCA pump in the atria and ventricles.
Babu G.J. et. al., PNAS 2007 104 (45) 17867-72.
Sarcolipin (SLN) is a small peptide found in the sarcoplasmic reticulum of skeletal muscle. It is predicted to contain a single hydrophobic transmembrane alpha-helix. Fluorescence emission spectra for the single Trp residue of SLN suggest that SLN incorporates fully into bilayers of dioleoylphosphatidylcholine, but only partially into bilayers of phosphatidylcholines with long (C(22) or C(24)) fatty acyl chains. The fluorescence of SLN is quenched in bilayers of dibromostearoylphosphatidylcholine, also consistent with incorporation into the lipid bilayer. SLN was reconstituted with the Ca(2+)-ATPase of skeletal-muscle sarcoplasmic reticulum. Even at a 50:1 molar ratio of SLN/ATPase, SLN had no significant effect on the rate of ATP hydrolysis by the ATPase or on the Ca(2+)-dependence of ATP hydrolysis. However, at a molar ratio of SLN/ATPase of 2:1 or higher the presence of SLN resulted in a marked decrease in the level of accumulation of Ca(2+) by reconstituted vesicles. The effect of SLN was structurally specific and did not result from a breakdown in the vesicular structure or from the formation of non-specific ion channels. Vesicles were impermeable to Ca(2+) in the absence of ATP in the external medium. The effects of SLN on accumulation of Ca(2+) can be simulated assuming that SLN increases the rate of slippage on the ATPase and the rate of passive leak of Ca(2+) mediated by the ATPase. It is suggested that the presence of SLN could be important in non-shivering thermogenesis, a process in which heat is generated by hydrolysis of ATP by skeletal-muscle sarcoplasmic reticulum.
Smith WS et. al. Biochem J. 2002 Jan 15;361(Pt 2):277-86.


